
BMS Cruiserath Pumped CIP: Chromatography Case Study
Introduction The Clean in Place (CIP) procedure for a chromatography column ensures thorough cleaning without disassembly. The process begins by flushing the column with deionised water to remove residual product....

Cascaded PID Control in the Autoclave Industry: Precision for Sterilisation Success
In the autoclave industry, precision and reliability are paramount. Autoclaves, used for sterilising equipment in medical, pharmaceutical and industrial settings, must meet rigorous standards to ensure the complete elimination...
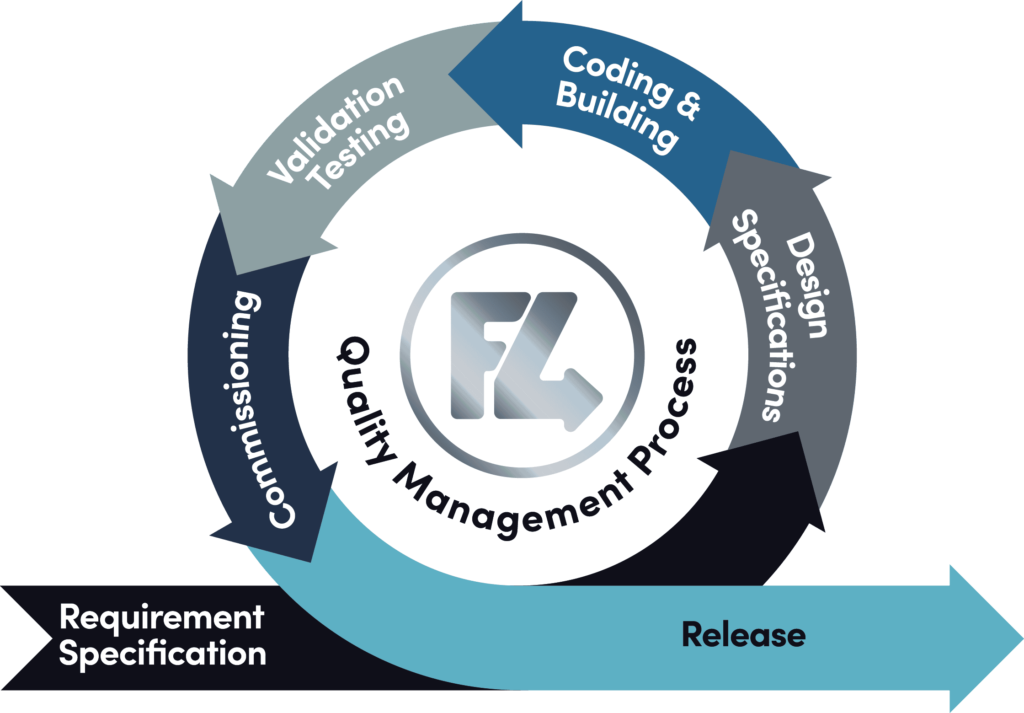
Quality Management Process – Feed4wards Approach
Here at Feed4ward we’re a process led company and our quality management process insures we achieve quality every time, providing a high level of validation before implementation to ensure automation...

GAMP5 Guidelines & How It Influences Software Design
When it comes to developing software, the chosen method of design can have a significant impact on the success and efficiency of the project. Different software design methods offer unique...
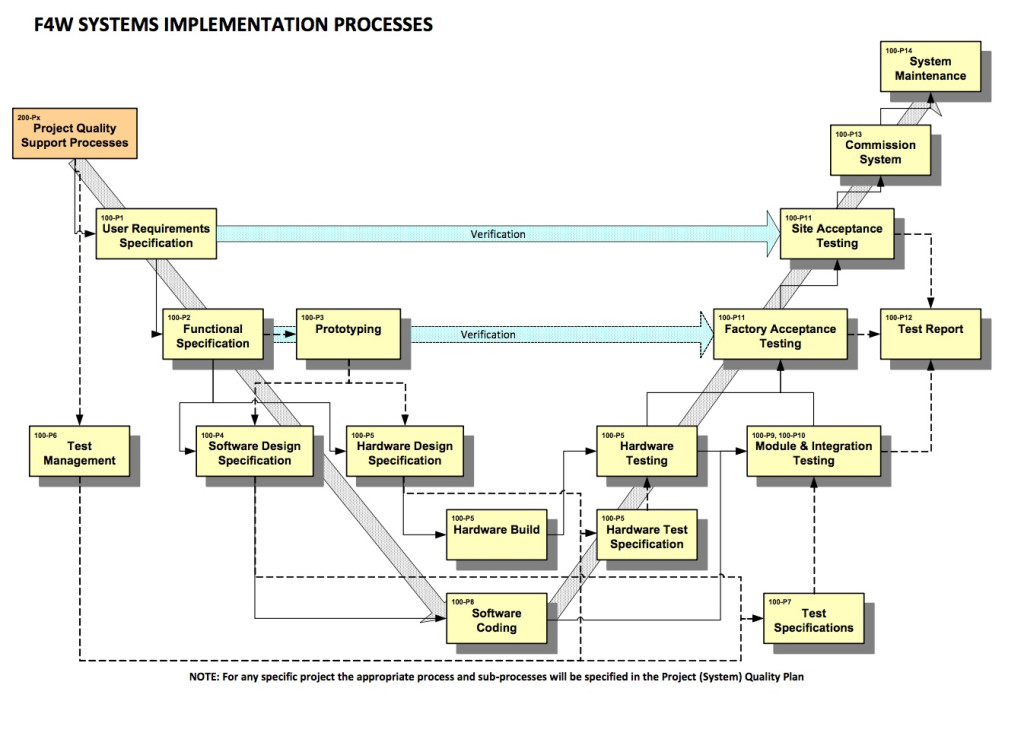
The importance of using the GAMP®5 guidelines for your QMS
If you’re in the business of Control and Automation, you’ve probably heard people talk about GAMP. You’ll know that it’s ‘some sort of quality thing for pharmaceuticals’. But what’s so...

Is GAMP®5 compatible with agile development?
As engineers and project managers, we like certainty. Detailed clear requirements, defined up-front, against which we can design, cost and plan. However the modern world is a dynamic, fast-changing place....