GAMP5 Guidelines & How It Influences Software Design

When it comes to developing software, the chosen method of design can have a significant impact on the success and efficiency of the project. Different software design methods offer unique advantages depending on the project’s requirements, complexity and risk.
It is also important to apply the right design approach to the specific demands of each project. Highly regulated industries such as the pharmaceutical industry, require rigorous design and validation methods to ensure conformity with regulatory standards.
Given our focus on solutions for the pharmaceutical industry, we frame our software design around GAMP5 (Good Automated Manufacturing Practice) guidelines. GAMP5 provides a structured, risk-based approach to guarantee that the software we develop:
- Meets the highest standards of safety
- Has measurable data integrity
- Is traceable to user requirements

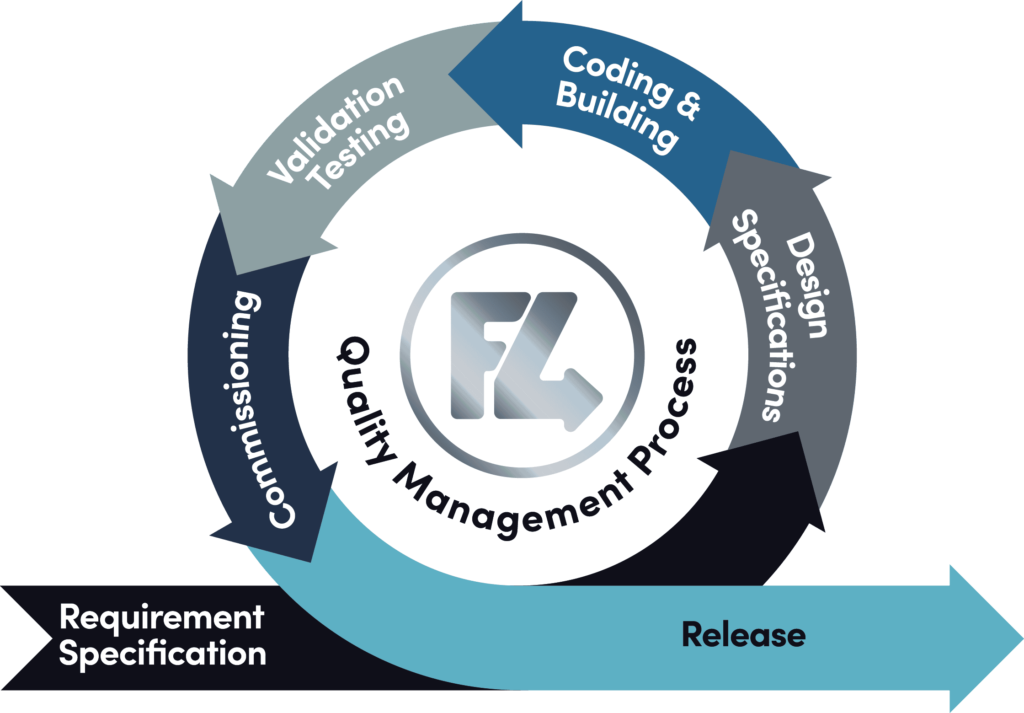
At Feed4ward, we use the V-Model for GAMP-compliant software design. The V-Model is well-suited for projects that require thorough validation and traceability. This model pairs each phase of development with a corresponding validation activity.
When a system is designed, validation tests are created ensuring every requirement is thoroughly verified before moving to the next stage. This design approach ensures that no steps are skipped, and any potential risks to product quality or safety are addressed early.
GAMP5 differentiates between off-the-shelf (COTS) software and custom-built applications. In a typical environment, COTS software is often integrated quickly with minimal adjustments. Although fully custom software (Category 5) requires end-to-end validation, from design through to final implementation. This involves far more detailed testing, risk assessments and documentation, than you would usually find in a non-GAMP software project. In typical software projects, data security is important, but the level of scrutiny often depends on the industry.
Data Integrity
For GAMP5, ensuring data integrity is a fundamental part of the design process. Since regulatory bodies like the FDA and EMA require that data related to product manufacturing be accurate, reliable, and traceable. Therefore in GAMP software design, audit trails are a critical feature that must be designed into the system from the very beginning. This means the software needs to be built with user role-based access controls, tamper-proof logging, and timestamped actions. Actions must be attributable to a specific user and traceable throughout the system’s lifecycle.
Data recovery and backup are standard in most software systems, but GAMP5 adds an extra layer of validation to ensure that data integrity is preserved during backup and restoration processes. This can mean designing more complex redundancy features and backup verification protocols to ensure that no data is lost or corrupted. In contrast, non-regulated software systems often do not require such controls when data accuracy and recovery are not critical.
A GAMP5-driven design requires that every requirement, feature and change be traceable from concept to deployment. This often means using traceability matrices that map out how each requirement is implemented, tested, and validated. At Feed4ward, every design decision is well- documented and impact assessed for how changes in one area might affect the system functionality elsewhere.
While agile methodologies focus on rapid iterations and continuous deployment, that can lead to multiple changes being released in quick succession. Under GAMP5, iterative development in high-risk areas translates to repetition of testing and validation, which can lead to delays in the project.
Summary
In summary, compared to typical software design approaches, GAMP5 imposes a unique, risk-based, and highly structured methodology. This prioritises regulatory compliance, patient safety and product quality, above all else. This has a profound impact on how software is designed, tested, and validated.
At Feed4ward, our commitment to GAMP5 ensures that our software solutions not only meet industry standards, but also enhance project efficiency. Whilst ensuring a high level of trust in every system we deliver.
Contact us to discuss your automation project.